Abstract
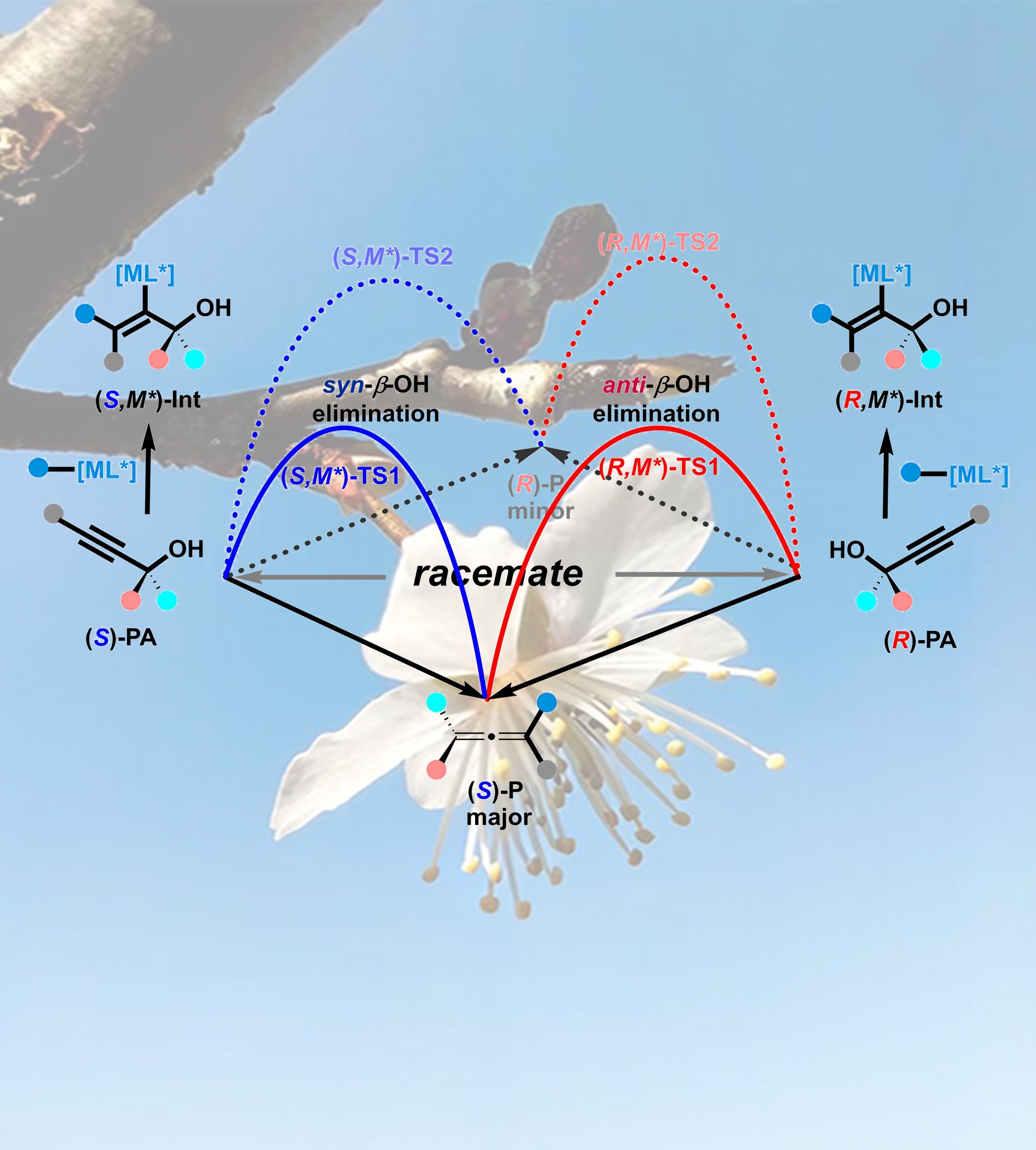
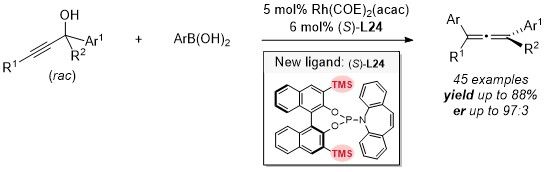
Optically active compounds are abundant in nature and would be of fundamental interest for organic, medicinal, and material chemistry. Catalytic transformation of readily available racemic compounds into same value-added enantiomeric products is one of the most efficient and cost-effective protocols. Herein, an SN2’-type enantioselective reaction of racemic propargylic alcohols with aryl boronic acids (both are readily available) under a rhodium catalyst empowered by a new phosphoramidite-olefin bidentate ligand affording tetrasubstituted enantioenriched allenes has been unveiled. A pair of diastereomeric vinylic rhodium species with an allylic hydroxyl group has been identified and characterized by ESI-MS and TWIM-MS. Based on the X-ray diffraction data of the catalyst and other mechanistic studies, it is confirmed that the reaction is a conceptually new approach of parallel path enantio-convergent transformation, which is different from the classic dynamic kinetic resolution and dynamic kinetic asymmetric transformations.
PPET